|
12/7/2023 0 Comments C2 molecular orbital diagram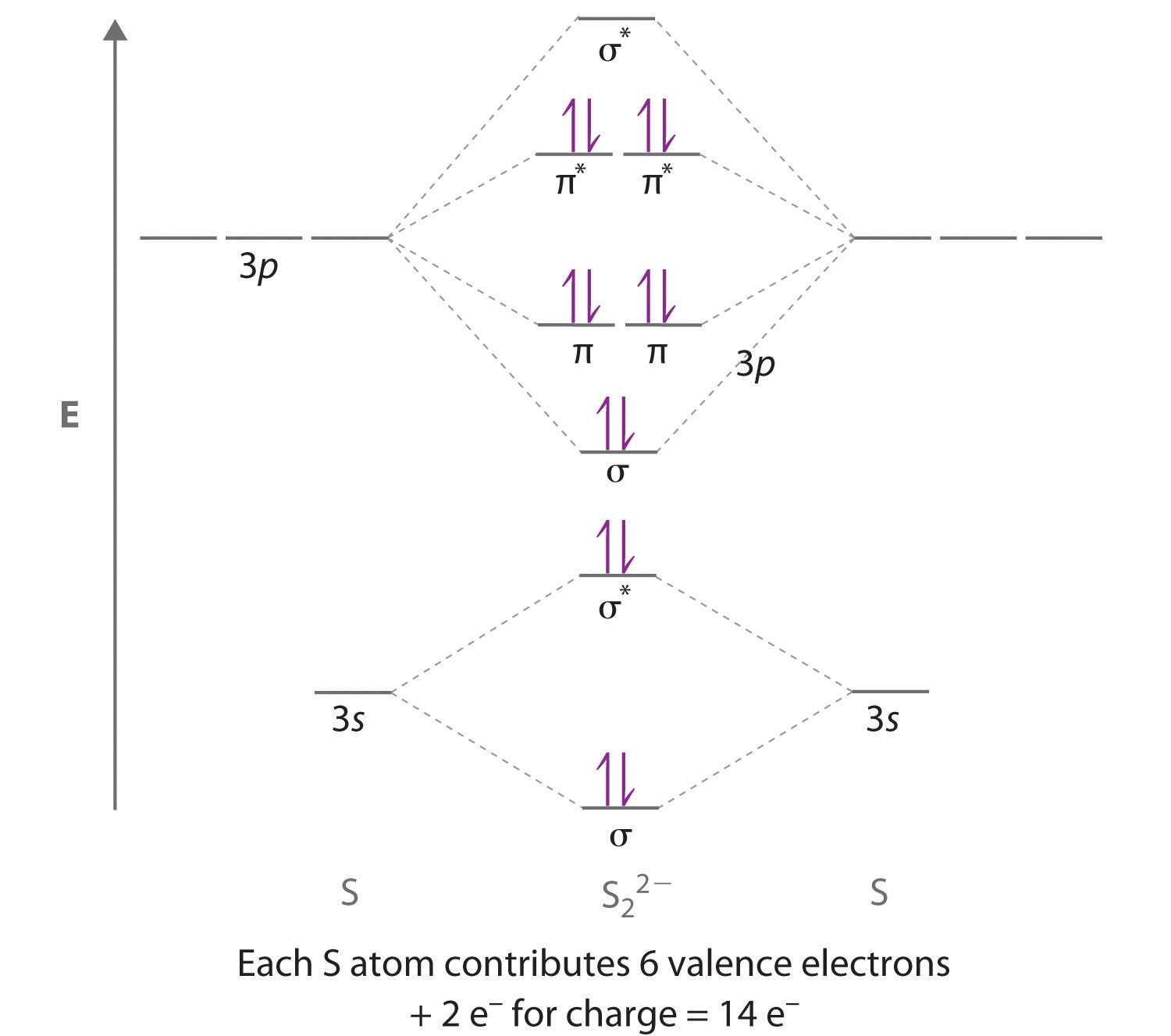
Adopting the MO diagram for O2, theorbitals of C2. 
OF has 14 valence electrons, four in the 2p orbitals (see the diagram in the answer to Problem 5.9). Nitrogen is the last element in the second period for which the diminishing effects of the orbital mixing are still significant enough for the energies of the #sigma_g(2p_z)# to still be higher than the #pi_(u(2p_x))# and #pi_(u(2p_y))# in energy.įrom #"O"_2# through #"Ne"_2#, the #sigma_g(2s)# and #sigma_g(2p_z)# are too far apart in energy to interact, so the orbital mixing effects are no longer as significant.Īt that point, the #sigma_g(2p_z)# has crossed the #pi_(u(2p_x))# and #pi_(u(2p_y))# orbitals in energy, so the orbital ordering "switches" and these diatomics have a #sigma_g(2p_z)# MO lower in energy than the #pi_(u(2p_x))# and #pi_(u(2p_y))# MOs. Molecular Orbital Diagram for C2 Carbon has 4 valence electrons, i.e.there are 8 electrons in MOs. The latter do not possess C2 rotation axes coincident to the infinite-fold rotation axis of the orbitals on the basis of the change in wave function sign upon crossing the nodes on the bond axis. Therefore, the effects of the mixing become less and less significant from left to right. This also causes a large jump in energy in the 2p. Notice how the from the 2p behaves more non-bonding like due to mixing, same with the 2s. This is the reasoning for the rearrangement from a more familiar diagram. This is often shown for the second-period elements.įrom #"Li"_2# to #"N"_2#, we would actually see a trend of the #sigma_g(2s)# decreasing in energy (faster than) the #sigma_g(2p_z)# decreases in energy the effect of orbital mixing decreases as the #sigma_g(2s)# and #sigma_g(2p_z)# get farther and farther apart in energy. With nitrogen, we see the two molecular orbitals mixing and the energy repulsion. Which is the most stable molecule Which molecule (s) is/are paramagnetic Draw the molecular orbital diagrams for C2-, C2, and C2+. Question: Draw the molecular orbital diagrams for C2-, C2, and C2+. It is analogous to the atomic orbital energy diagram (which goes 1s, 2s, 2p, 3s.). The middle of the diagram is just the molecular orbital energy diagram. Not surprisingly, this effect is called orbital mixing, and the result of it enhances the bonding with additional electron stabilization. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. What we see here is a molecular orbital interaction diagram. In #"N"_2#, the #sigma_g(2s)# and #sigma_g(2p_z)# molecular orbitals are compatible (they are both symmetric with respect to infinite rotation and inversion), AND they are close enough in energy (generally within #pm "12 eV"#), so they mix. Bond order 1 2 ( number of bonding electron - number of antibonding electrons) 1 2 ( 10 - 4) 1 2 × 6 3. It shows electrons in both bonding and anti-bonding molecular orbital. Strategy Follow the LCAO method described in Box 4. 209 in Chemistr圓) Write an expression for the wavefunction of the bonding orbital in a molecule of LiF. So, without further ado, let us take a look at the. WE 4.6 Equations for the molecular orbitals in HF (on p. Notice that all of the above are diatomic molecules. The Question and answers have been prepared according to the JEE exam syllabus. This effect is still barely present on #N_2#, so its #sigma# bonding MO is still higher in energy than its #pi# bonding MOs. The molecular orbital diagram has molecular orbital energy level at centre and is surrounded by atomic orbital energy level. Here is the list of MO diagrams shown in this gallery: O2 molecular orbital diagram. Molecular orbital energy diagram of C2 for JEE 2023 is part of JEE preparation. The lowest energy unoccupied molecular orbital is $ 2p\sigma $, so that is where extra electrons will be added.Because from right to left, the orbital mixing interaction between two compatible orbitals of similar energies decreases the energy of the lower- energy orbital (relative to what it would be without mixing) and increases the energy of the higher- energy orbital (relative to what it would be without mixing). 
The diagram below shows the two $ 2p\pi $ orbitals, let's say $ 2p\pi x $ and $ 2p\pi y $, are the highest energy occupied molecular orbitals. It will be added to lowest energy unoccupied molecular orbital/lowest unoccupied molecular orbital, LUMO, that follows that the highest energy occupied molecular orbital/highest occupied molecular orbital, HOMO.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
